I run Science Crazy Clubs in Ipswich and we've been having so much fun learning hands on. I sometimes even share a few of the projects we do on the Science Crazy Blog.
The last Science Crazy club was all about how catapults worked and so we made some fun catapults and had fun testing them. The most recent blog I have shared on Science Crazy is this fun Catapult made from Lolly pop sticks and elastic bands!
The link to the instructions for how to make one yourself can be found by clicking on the photo. All you'll need are lolly sticks and elastic bands and find something to propel... a marshmallow will do! Do let me know how you got on if you try it - I love to hear from my readers!
This is my daughter playing with her own catapult. I love to make our home education as hands on as possible as learning through play is the best way, especially through their younger years I think.
Friday 26 January 2018
Wednesday 7 September 2016
Science: Lego Soil Layers
So, I was inspired by a photo I saw floating around the web the other day and just had to fit it into our learning on Monday.
My 6 and 8 year old loved it so I thought I would share so you can have a try too!
If you like you can follow this activity up with another more scientific version, called: What's in Soil? that can be found by clicking on the photo below...
My 6 and 8 year old loved it so I thought I would share so you can have a try too!
If you like you can follow this activity up with another more scientific version, called: What's in Soil? that can be found by clicking on the photo below...
Thursday 25 August 2016
STEP by STEP How to make Slime
In this post I am going to show you how to make slime that is safe for young children to make and play with, the other than needs adult assistance.
- GOGGLES
- PVA GLUE
- Starch Spray
- 2 CUPS (clear cups or glasses are better as the children can see what's
happening) - FOOD COLOURING (optional but adds to the fun)
ALWAYS WEAR GOGGLES WHEN DOING SCIENCE EXPERIMENTS. It's
important to practice. Also, wear aprons or get yourself a lab coat
and be like a real scientist! It will protect you and your clothes
from being damaged. This silly putty can stain and ruin,
clothes, carpets, curtains etc.
STEP ONE
Put about 30 ml of PVA glue in a cup, mix a little food colouring in.
STEP TWO
In another cup spray the starch spray until you have about 40ml of liquid starch settled in the container.
In another cup spray the starch spray until you have about 40ml of liquid starch settled in the container.
STEP THREE
Add the liquid starch to the PVA and colour and stir until it all binds together.
Spaghetti is a very useful way of showing how and why polymers act the way they do.
Spaghetti is made of long strands very much like the molecular structure of a polymer.
When you boil up the spaghetti it runs like water, but then you take it out of the water and the starch makes it sick together and feel a bit sticky.
Leave it a bit longer and you can bounce it! This is due to the starch in the pasta.
Spaghetti is made of long strands very much like the molecular structure of a polymer.
When you boil up the spaghetti it runs like water, but then you take it out of the water and the starch makes it sick together and feel a bit sticky.
Leave it a bit longer and you can bounce it! This is due to the starch in the pasta.
Why is spaghetti such a good example?
It is a great example because the molecules in the crazy putty are a linear polymer. Which means the molecules are all in a line rather than branching. Linear in this sense does not mean a straight line but one like cooked spaghetti that curls about.
It is a great example because the molecules in the crazy putty are a linear polymer. Which means the molecules are all in a line rather than branching. Linear in this sense does not mean a straight line but one like cooked spaghetti that curls about.
Something else I like to do when there are plenty of people about to join is, is to make a model of a polymer using HUMANS!!
Ask at least 5 to stand in a row holding hands. Now they are like a linear polymer molecule. It's that simple.
You can try adding the PVA glue to the starch liquid instead of the start to the PVA glue...
You can try adding the PVA glue to the starch liquid instead of the start to the PVA glue...
OTHER POSTS THAT TEACH KIDS ABOUT POLYMERS...
HOW TO MAKE MAGNETIC PUTTY - and a video of how magnetic putty works!
First posted 5 August 2011 - Updated 25 August 2016
Thursday 14 July 2016
Day 4 of the Butterfly project - Maths: STEP by STEP caterpillar geometry
I've been trying to think of ways to get maths into our butterfly project as much as possible and came up with this project.
As we learnt yesterday, caterpillar eggs come in various shapes sizes, some are tubular in shape, some round and others oval, so in this project we make a caterpillar egg and the turn it into a caterpillar... Just for FUN! Here's how we made ours...

These two bigger triangles are isosceles and the smaller ones are right angle triangles.
Maths Fact: the three angles inside a triangle always add up to 180 degrees!
STEP FIVE
Mark 1 cm down from the inside line of the small rectangle and 1cm up from the inside line of the small rectangle and then join the dots to the corners of the newly made isosceles triangles to make smaller right angle triangles, like so...
STEP SIX
Now colour it in!
We were thinking of 'The Very Hungry Caterpillar' by Eric Carle, when we made ours so we coloured the semi circles in red, the large rectangle in green and the triangles orange.

STEP SEVEN
Then we cut it out carefully.
If you like, before you rearrange the pieces and put them back together, you can draw a background on an A4 piece of paper. Something like this...

Then stick your shapes on the picture to make a caterpillar shape like this...

What we learnt from this project:
As we learnt yesterday, caterpillar eggs come in various shapes sizes, some are tubular in shape, some round and others oval, so in this project we make a caterpillar egg and the turn it into a caterpillar... Just for FUN! Here's how we made ours...

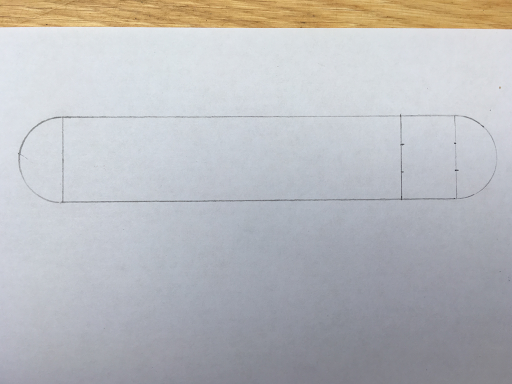
STEP ONE
Draw a rectangle. Make it 3cm by 14cm.
To ensure the angles of your rectangle is right use a protractor. All the angles are meant to be 90 degrees.
Maths fact: All the angles on a quadrilateral adds up to 360 degrees
STEP TWO
Using a compass set it to 1.5cm like this...
STEP TWO
Using a compass set it to 1.5cm like this...
Then draw a semi circle at both ends of your rectangle.
STEP THREE
STEP THREE
Now measure two centimeters in from one end of the rectangle we made in STEP ONE and then draw a line to make a small rectangle within it like this...

STEP FOUR
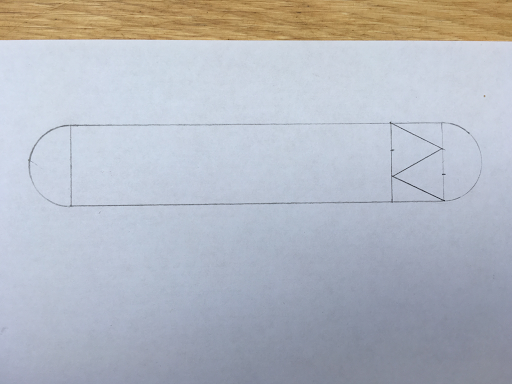
Measure and mark with a pencil 2cm from the top of the inside line of the small rectangle and 2cm from the bottom of the line joined to the semi circle. Use these marks and the corners of the small rectangle to make triangles like in the photo below.
Measure and mark with a pencil 2cm from the top of the inside line of the small rectangle and 2cm from the bottom of the line joined to the semi circle. Use these marks and the corners of the small rectangle to make triangles like in the photo below.

These two bigger triangles are isosceles and the smaller ones are right angle triangles.
Maths Fact: the three angles inside a triangle always add up to 180 degrees!
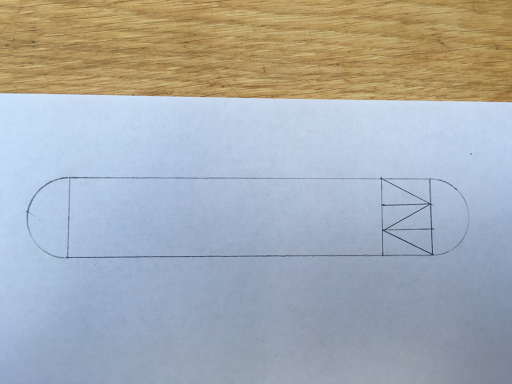
STEP FIVE
Mark 1 cm down from the inside line of the small rectangle and 1cm up from the inside line of the small rectangle and then join the dots to the corners of the newly made isosceles triangles to make smaller right angle triangles, like so...

STEP SIX
Now colour it in!
We were thinking of 'The Very Hungry Caterpillar' by Eric Carle, when we made ours so we coloured the semi circles in red, the large rectangle in green and the triangles orange.

STEP SEVEN
Then we cut it out carefully.
If you like, before you rearrange the pieces and put them back together, you can draw a background on an A4 piece of paper. Something like this...

Then stick your shapes on the picture to make a caterpillar shape like this...

What we learnt from this project:
- Counting
- Measuring
- Shapes
- Angles
- Practicing using a compass and ruler
Tuesday 12 July 2016
Butterflies day 2: Egg on Leaf craft
So today wasn't all about butterflies (only 3 days to go!) but, after our maths and English workbooks, we did do a quick craft for our scrapbook to keep the theme going...
We made a fun craft painting the leaves, then drawing and colouring tiny eggs using photos we found on the internet. It was amazing to see all the different shapes and colours of butterfly eggs there are.

WE cut the egg drawings out and using glue stuck them to the leaf. I talked to them about how the female butterfly, when she lays her eggs, secretes a special chemical that acts like a glue to secure her eggs to the leaf she chooses.
This was their finished egg on a leaf art...
We made a fun craft painting the leaves, then drawing and colouring tiny eggs using photos we found on the internet. It was amazing to see all the different shapes and colours of butterfly eggs there are.

WE cut the egg drawings out and using glue stuck them to the leaf. I talked to them about how the female butterfly, when she lays her eggs, secretes a special chemical that acts like a glue to secure her eggs to the leaf she chooses.
This was their finished egg on a leaf art...
Monday 11 July 2016
Butterflies day 1: Maths through art
We woke up this morning excited about the arrival of our caterpillars!

The first thing we did, even before getting dressed or eating breakfast was finding the hungry caterpillar puzzles!
Once we had eaten breakfast we started on our project. This weekend I popped to town when I was child free and found, in The Works, these lovely butterfly masks...

This morning they started to colour them in. We talked about symmetry familiarizing the younger two with the word that they will find in their maths workbooks.
So much fun.
They used there feet to make a giant one too that is now on my kitchen door...

Then we found out that we don't get the butterflies today. They are coming on Friday. Something I had miss read no doubt - I can be ditzy at times.
Still we continued to have fun. We made those carrot cakes inspired by Hugly and Friends book, and turned some into butterfly cupcakes. If you click on the photo (after 8pm tonight) it will lead you to the recipe!

We are going to spend the next few days preparing for our caterpillars instead, so as promised I will continue blogging about butterflies over the next few days but no photos of caterpillars until Friday now!

The first thing we did, even before getting dressed or eating breakfast was finding the hungry caterpillar puzzles!
Once we had eaten breakfast we started on our project. This weekend I popped to town when I was child free and found, in The Works, these lovely butterfly masks...

This morning they started to colour them in. We talked about symmetry familiarizing the younger two with the word that they will find in their maths workbooks.
We then got the paint out and experimented with other ways of using art to create symmetrical butterfly shapes! Just the simple old fashion method I remember from when I was small. A folded piece of paper, paint on one side squish it together - the sort of craft that never loses its magic!
So much fun.
They used there feet to make a giant one too that is now on my kitchen door...

Then we found out that we don't get the butterflies today. They are coming on Friday. Something I had miss read no doubt - I can be ditzy at times.
Still we continued to have fun. We made those carrot cakes inspired by Hugly and Friends book, and turned some into butterfly cupcakes. If you click on the photo (after 8pm tonight) it will lead you to the recipe!

We are going to spend the next few days preparing for our caterpillars instead, so as promised I will continue blogging about butterflies over the next few days but no photos of caterpillars until Friday now!
Sunday 10 July 2016
Book Review: Hugly and the Missing Carrots
The other day we received a special something in the post...


Hugly and the Missing Carrots!
It's a book written by two dads, Ian Hamilton and Sam Frith, their aim for this book, they say, is to encourage children to learn about nature and its surroundings, growing fruit and vegetables and cooking in which I think they have completely accomplished with their book which has been possible using crowdfunding.
It is such a lovely tale of love and friendship. Before we even began reading the book she noticed the special little envelope...
Wednesday 17 February 2016
Science: Sticky Static
Static electricity is an electric phenomenon that is well known. It is where charged particles are transferred from one place to another.
Here's something fun you can try when you are learning about static electricity and is great as an introduction to electricity.
When you rub a balloon against your hair or a jumper, charge transfer happens, this is when static electricity is made; when you rub a balloon against your jumper or hair, the balloon steals electrons (negatively charged particles) when this happens you leave the jumper, or your hair in a positively charged state and the balloon is negatively charged.
The balloon will be attracted back to the sweater because opposite charges attract like opposite pole attract with magnets.
The reason that the balloon will
stick to the wall is because the negative charges in the balloon will make the
electrons in the wall move to the other side of their atoms (because like charges
repel) and this leaves the surface of the wall positively charged. Because
opposite charges attract, the negatively charged balloon will be attracted to
the positively charged surface of the wall.
Something else you can try:
Try using different materials to negatively charge your balloon. Which material works best?
Scientific words learnt through this experiment:
- Static electricity
- Protons
- Electrons
- Negative charge
- Positive charge
Thursday 11 February 2016
Science: Experimenting with Copper Sulphate
First of all Heating copper...
When it is cooled down obviously,
you can clear away the copper oxide.
We could put an iron nail in it. What happens? The liquid turns from blue to green.
When an iron nail is placed in a copper sulphate solution, iron displaces copper from copper sulphate solution forming iron sulphate, which is green in colour.
Therefore, the blue colour of copper sulphate solution fades and green colour appears.
When we take an iron metal nail and dip it into a blue solution of copper (II) sulphate, firstly it changes the solution to iron sulphate which is green in colour. If you leave it the nail becomes covered with a reddish substance. This reddish substance is metallic copper. The equation for this is:
This post was first written 11/07/2012 revised: 11/02/2016
One of my friends who come along
to my science groups back in 2012, asked me if we could do some experiments on
heating materials. She said she had
tried heating a copper coin and couldn’t get it to spark green and didn’t know
why. Well, this is why it didn't produce that green flame...
If you heat a modern copper coin
it will go black, no green flame. This
is because you have formed copper oxide; the black formed during the heating of the coin in flame is the copper oxide.
Then I was left with the
question: how could I make a green flame using copper? How I did this was not using a copper coin But I did use copper sulphate.
Using a spoon and an alcohol burner I heated it and voila green
flames! Fab. Be sure that you’re in a ventilated area when
you do this.
What else can we do with Copper
sulphate? We could put an iron nail in it. What happens? The liquid turns from blue to green.
When an iron nail is placed in a copper sulphate solution, iron displaces copper from copper sulphate solution forming iron sulphate, which is green in colour.
Therefore, the blue colour of copper sulphate solution fades and green colour appears.
When we take an iron metal nail and dip it into a blue solution of copper (II) sulphate, firstly it changes the solution to iron sulphate which is green in colour. If you leave it the nail becomes covered with a reddish substance. This reddish substance is metallic copper. The equation for this is:
Fe(s) + CuSO4(aq) -> FeSO4(aq)
+ Cu(s)
This is what is known as a redox reaction, or otherwise known as a oxidation reduction reaction.This post was first written 11/07/2012 revised: 11/02/2016
Thursday 4 February 2016
Science: How to make glue from milk
This is a fun experiment, it does involve heat so you do have to be careful and have a responsible adult when carrying out this investigation.
As you can see in this photo the glue we made was strong enough to stick two boxes of eggs together and hold the weight of the six eggs in the bottom box! Pretty impressive!
As you can see in this photo the glue we made was strong enough to stick two boxes of eggs together and hold the weight of the six eggs in the bottom box! Pretty impressive!
SO...
For this scientific investigation I will take you through step by Step explaining the science behind it as we go. First you will need:
Monday 11 January 2016
Science: How to make Carbonic acid
When carbon dioxide is added to water, the gas adds acidity to the water. The reaction between the Carbon dioxide and the water is a synthesis reaction, sometimes known as a combination reaction, and creates an acid known as Carbonic acid.
In this post I will talk you through Step by Step on how to make Carbonic Acid and the Science of the reaction as we go...
Wednesday 11 November 2015
Wednesday 28 October 2015
Science: How to make a Geode
So this is how to make your own geode!
Ok, it's not a real geode but this simple but fun experiment can help kids understand they are formed. To be honest, how geodes are formed is still a phenomena but scientists have a couple of theories one of these theories is lava!
First here's how to make your own geode...
Ok, it's not a real geode but this simple but fun experiment can help kids understand they are formed. To be honest, how geodes are formed is still a phenomena but scientists have a couple of theories one of these theories is lava!
First here's how to make your own geode...
Wednesday 23 September 2015
Chemical Reactions: combustion of iron
In this experiment we are going to look further into what happens when the combustion of iron occurs...
The definition of combustion: The process of burning something.
Combustion is a chemical reaction.
Combustion is a chemical reaction.
Have you ever thought of what somethings weighs before and after it's burnt? Well in this experiment that's exactly what we are going to do!
When doing this experiment please be careful as it does involve fire. Ensure there is nothing like paper or cloth or anything else that can easily catch fire around. Also it is very important that you wear goggles for this experiment.
What you will need:
- A bunsen burner, or chefs torch - you could try an alcohol burner if you have one
- Steel wool - like you use to scrub the pans
- Tongs
- Weighing scales
- A metal baking tray
How to do this investigation...
Wednesday 16 September 2015
HEAT ENERGY: A Convection and Conduction experiment
This is very simple demonstration of how conduction and convection heat energy works. It involves fire which usually fascinates children.
For this experiment, you will need...
Subscribe to:
Posts (Atom)

















